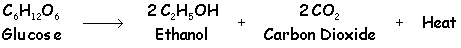
| Summary |
|
Ferment the mash/wash at a constant 25oC, using 1.5g/L of good yeast
suited to the wash. Use an airlock to let CO2 out but not let air in. Let the yeast settle out, and possibly even filter the wash before putting it into the still. |
| Tall fermenter | Short/wide fermenter
| Fermentation time: | 10 days | 8 days
| Final gravity: | 1.010 | 1.011
| Diacetyl (mg/L) | 0.350 | 0.060
| Clearing | poor | excellent
| pH at end point | 4.6 | 4.4 | |

| Temperature | Ethyl acetate(mg/l) | Amyl acetate(mg/l) |
| 60F (15.5C) | 16 | 0.5 |
| 68F (20C) | 26 | 2.0 |
| 75F (24C) | 53 | 4.0 |
| High Alcohol Fermentation 15-20 +% v/w | |
| K-1or L 2226 Yeast Strain | 6# / 1000 gallons |
| Vi-A-Dry Inactive Yeast 2133 | 2# / 1000 gallons |
| Fermaid K | 2# / 1000 gallons |
| Di Ammonium Phosphate (DAP) | 4# / 1000 gallons |
| http://homedistiller.org This page last modified |