Home Distillation of Alcohol (Homemade Alcohol to Drink)
Stills that Aren't Stills
It is possible to collect alcohol from a wash, without using a full-blown still.
They use common kitchen items, and takes seconds to construct / dismantle (hmmm... say the Yanks).
My only concern is that there is no way to remove & discard the
methanol/fusels etc - so you're in for one hell of a hangover if starting
with fruits high in pectin. These methods don't produce a great product (require a second redistillation through
the equipment again, or use carbon to clean it up), and are low
in % alcohol, but they are an alternative technique, and are
better than nothing.
Slow Evaporation
Johan writes :
I made a very simple "still" once. It worked like this:
In a 25 liter bucket i put a 10 liter bucket filled with mash.
I added a aquarium heater in the small bucket and had a constant
temperature of 40 degrees celcius.
Then i put the lid on the 25 Liter bucket and waited (and waited and
waited). The air inside the 25 L bucket became saturated with alcohol/water and
condensed on the 25 L wall.
24 H later about 300-400 mL 40% alcohol was found in the bottom of the 25L
bucket.
The quality was very nice! much better than a average fractional still.
To make this work you have to have strong mash, about 20%
This idea has now been written up in more detail at http://www.amazingstill.com, and
there is a 600kB PDF file all about it, how to make it etc :
Amazingstill.pdf.
It has no moving parts, is very cheap and simple, no odour, no cooling required, and will produce
around 1/2L a day, and requires no attention - can be left alone to do its work slowly over time.
There is some suggestion that evaporation at low (45-50C) temperatures results in less fusels
than from higher (80-90C), so that the spirit is very clean and smooth.
Johan adds ..
This still is for people that wants a simple but effective apparatus.
Some parts that is left out in the text and worth mentioning is the different
kind of aquarium heater you can use.
1. One of the best one is the Jäger heater SH not adjustable 100W, you can
find it in www.aquaristikshop.com (search for jäger
on the site) It is cheap and it has a thermostat that is set on 47 degrees C
(it thinks it runs dry over that and shuts down). Perfect for our needs, just plug it in and
you get the right temperature. Maybe partyman will sell this one as well, we'll see.
Unfortunately Jägers green wire can't withstand alcohol. In time it
will break down. Thread the wire in a hose and seal with glass silicone
and a hose clip.
2. Modification of ordinary aquarium heater: Some have a lock that makes it impossible
to get over 32 C. Remove the plastic lock and you can have 50 C if you wish.
3. Those that can't be modified with lock: 99% of all aquarium heaters have a
bimetal thermostat, open the heater and force the metal down with a ball of paper
so that the thermostat never shut down the heater. Adjust the effect with a dimmer.
Riku has made a neat modification to the basic model, with a hot air gun he changed
the shape of the lid to a dome so all condense was collected. Another way is to tilt
the bucket. This is useful if you want to use it as a prestill your mash before your
ordinary still. That way the quality is improved.
Bucket Still
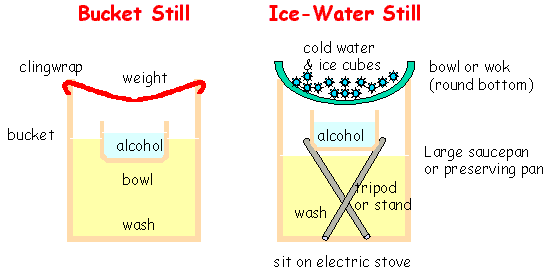
The Bucket Still is the simplest.
Float a bowl in the wash, cover the lot with a layer of plastic or clingwrap.
Sit something heavy on the clingwrap so that any condensate that forms on the wrap
will run down to the middle, and drip into the bowl. Leave the bucket etc
somewhere warm. This process will take a long
time to collect very much at all, and will be of quite low strength,
but is probably better than nothing. Easily adapted for use in your cell.
Ice-Water Still
This concept can be beefed up a little, by heating the wash and cooling the vapour
more efficiently (the Ice-Water Still). In
"Brewing Real Schnaps Without a Still" (sorry, dead link:
http://olliver.family.gen.nz/schnapps.htm) Vik describes
a simple method using just a bowl & a saucepan, to make schnapps.
To prevent the collected alcohol from re-evaporating, it would be wise to support
(ie on a tripod ?) the collection dish above the wash, so that it remains cool(er)...
Vik tells me that the bowl floating in the mash doesn't get as hot as
I'd thought because both the alcohol evaporating from it, and the cool
liquid dripping into it keep it at a
temperature lower than the mash, and that the continuous circulation
of alcohol might even improve the flavour. He can get results >50%
alcohol by volume with a single pass, though this does need a stiff brew to start with.
Just remember that your final product is highly flamable, so take care when making
it on the stove-top.
Jack describes how to use it for whisky or schnapps ..
After looking over the "double
distillation for whiskey" method on your site, I did some playing around with
a large (20 litre) version of the ice water "wok for a condensor" design.
Because the collected distillate is sitting on a tripod made of copper tubing
(nothing special- just three pieces of thin tubing cut to fit into a tripod
and support a bowl above the mash) the distillate is collected very hot-
making hydrometer readings impossible (yes, you could probably adjust for
temperature, but the stuff is still steaming- it would break the proof
hydrometer). To get around this problem, I figured out a method of
distilling spirit by measuring the amount collected. Here is how it works:
1.start with a mash of a 10%abv, 20 liters total volume, run it through your
potstill until you collect the first third. Volume collected= 6,666.6ml
(yes, I am this precise in my collection measurements) The temperature you
run this first run at is pretty unimportant. On the second run, use the
lowest power setting that you have.
2. load the 6,666.6ml of "low wines" into your potstill and run it on it's
lowest possible power setting. Collect the first 100ml of spirit as "heads"
(don't drink them! use them to start the bar-b-q). Now, collect the next
1,641.6ml of spirit that comes out of the still. With a starting mash of
10%, following these directions, this spirit should come out at 80%abv (at
least with my ice-water/wok still it does- suprising since this is the least
efficient still design I've ever worked with- make sure to use the LOWEST
power setting on your stove). This is your finished whiskey- cut and age it
as you wish.
3.Collect the next 580.6ml of spirit that comes out of the potstill as
"tails". These are to be added to the mash of your next batch- this
separation point ensures no heavy alcohols get into the finished spirit, but
no alcohol is wasted.
That's it- out of a 10%abv mash on the first run collect 1/3, on the second,
collect the heads, then the next 1/4, then collect the tails (enough to total
1/3 collected out of the second run). This method gives me 4 bottles of
great malt whiskey at 40%abv when I'm finished, along with a little more than
a half liter of tails for the next batch (you can also save the tails, and
when enough has been collected, run out the first 1/4 of the total as a
finished whiskey- it should be very good- I haven't collected enough tails to
try it yet). This system works just as well for brandy and schnapps. and is
usefull when you don't have a nice storebought still- hell, it doesn't even
require a thermometer! Using this system, the ice water/wok still can be
made with a 20liter aluminum stockpot and a glass bowl to work as the
condensor, and it will work just as well as any normal potstill.
The bowl I use for the lid/condensor is glass and it lets me see into the
collection bowl to check if it's full- if it is- siphon out the water, remove
the bowl, get a face full of steam, then use an oven mitt to grab the lip of
the bowl and pour the contents into a pyrex graduated cylinder- make a note
of the volume, then pour into an old 5 liter wine jug (1 of 2) that are
labeled "low wines". I can only tell when the collecting bowl is full-
because of the water I'm looking through (in the bowl) any "full line" marks
I put on the collection bowl would not be visible.
I have run across a
picture of an old still that had the same tall pot with a tripod supporting a
collection bowl (made of copper), but coming out of one side of the
collecting bowl (and through the wall of the still "body") is a tube that
allows one to collect the distillate without opening up the still. I would
like to try this- no time. Making a funnel out of copper sheet, and
attaching a length of tubing to the funnel's narrow end, then feeding the
tube through a hole in the still body (maybe stopping any leakage with lots
of teflon tape) so the tube extends out one or two feet and drops the
distillate into a cup would be nice, elegant way of avoiding a constantly
steam-burned hand and face.
p.s. when looking for a bowl to work as the
condensor, make sure it has a narrow base, or the distillate will fall off of
it and miss the collection bowl.
Solar Still
These are both based on the solar still, sometimes used in desert
regions. Patrick explains:
When I was a boy growing up in Arizona, USA, I learned how to make a desert
survival solar still. I often thought that it might work to distil whiskey. I
have never tried it with whiskey but it works to purify water. You dig a hole in the ground as big as your plastic sheet or nylon poncho
will cover. You then put chopped cactus plants into the hole. If there is damp
earth in the hole then no cactus needed. Put a container like a can or bowl in
the bottom of the hole right in the middle. You then place your plastic or nylon
poncho over the hole and place rocks around the edge of the covering until it is
secure and forms a bond over the hole. You then carefully put a stone in the
middle of the plastic sheet, causing the sheet to have a low spot right over the
container. This solar still will produce drinking water out of the desert soil. If
you were to dump mash into the hole before covering it with the plastic
sheet this should produce alcohol. I'm not sure but this may be how they made
mescal (tequila) in the old days. I don't know if this would work in cooler climes under 100 deg F.
Freezing
Another technique is to freeze the wash,
and separate the ice (water) from the
alcohol. Freezing appears to fall somewhere in-between the legal
issues for many areas (eg can you really stop your Applejack
from freezing if its left outside ?). Sort it out with your
own authorities (or just keep your freezer contents personal).
One thing to be aware of is the the more alcohol that is in the wash,
the cooler it will need to be before it freezes ...
Disolving (or mixing) something into a liquid will make the freezing point
go lower than when it is pure (see the full theory at
http://www.chem.vt.edu/chem-dept/dillard/1074Lecture27.pdf)
dT = Kf m where Kf(water) = 1.86
Adding ethanol to water will therefore depress the freezing point by 1.86 C per molar
conc present. Doing the translation between molar and regular % by volume gives ...
| 5% abv | 1.6 mol% | 3 C lower
|
| 10% abv | 3.3 mol% | 6 C lower
|
| 20% abv | 7.2 mol% | 13 C lower
|
| 30% abv | 11.7 mol% | 22 C lower
|
| 40% abv | 17.1 mol% | 32 C lower
|
| 50% abv | 23.6 mol% | 44 C lower
|
This explains why beer (at 5% abv) will freeze for ya in the freezer, wine will
need it to be a bit colder, but frozen schnapps or vodka won't normally be possible.
Scott adds more details ...
from http://users.stargate.net/~mshapiro/cfoot.html ..
Another method, known as fractional crystalization, is done by
inverting the process and freezing the beverage instead of boiling it.
This works for very similar reasons to that of normal heat distillation,
namely, the differential in freezing points of the two liquids involved.
Water freezes at a temperature of 0 C, while ethyl alcohol does not
freeze until reaching -114 C.
This allows
the water to be frozen out of the liquid, leaving behind the ethyl
alcohol, as well as the other alcohols and esters. This produces a drink
of a rather different character from heat distillation, as it contains
everything except water, while heat distilled beverages leave everything
behind except alcohol. Note also that simply lowering the temperature to
0 C will not produce an increase in alcoholic strength. The temperature
required for this process is in the range of -15 C and below, but must
vary, much as the diurnal cycle naturally does.
This allows
crystals of ice to form as the temperature drops. As the temperature
rises slightly the alcohol will drain out of the crystals so that when
the temperature again goes down and more crystals of ice re-form they
are purer crystals of water, containing less alcohol. As this process
repeats itself the solution will gradually work its way toward the
alcohol concentrations listed in the following table which is adapted
from a chart on page 102 of the book, Wines & Beers of Old New England:
| Temperature (F/C) | % Alc.
|
| 10 / -12.2 | 8
|
| 5 / -15.0 | 11
|
| 0 / -17.8 | 14
|
| 10 / -12.2 | 8
|
| -5 / -20.6 | 17
|
| -10 / -23.3 | 20
|
| -15 / -26.1 | 24
|
| -20 / -28.9 | 27
|
| -25 / -31.7 | 30
|
| -30 / -34.4 | 33
|
There is an excellent account of the history of using freezing at
Whisky on the Edge by
Alex Kraaijeveld.
I've just tried making some myself, with no success. Poured 1.5L of beer into a plastic
container, and put it in the freezer. When frozen, I broke it open. The trouble was though
that it appears that it all froze too fast - any alcohol there is trapped within the ice matrix -
there was no clean "core" of alcohol. I think that if this is to work, you will have to freeze
it VERY slowly, so that the alcohol has time to diffuse its way ahead of the freezing ice front.
Raj tells me though that to get a good clean separation ...
The trick with freezing is to partially thaw and refreeze a few times to promote
larger pieces of ice--the frozen cylinder with a heart of alcohol is the
consequence of diurnal cycling in a barrel over weeks.
Jack tells me though ..
Don't bother trying to get a frozen heart of alcohol- it's not really needed. Just
fill a gallon (4liter) milk jug half full of your beer/wine, let it freeze
solid in the freezer, then get a one quart (1liter) wide mouth jar, and stand
the milk jug upside down in it. It will take about four hours or so, but
eventually as the ice slowly melts (don't apply any heat), the jar will fill
up, and you will have just about doubled the alcohol (by cutting the volume
in half). using a 10% wine this method will turn a half gallon into a 17%
quarter gallon. I have not tried sticking this 17% wine back in the freezer
to see if I can get any more ice out of it, but it's worth a try. It also
seems to be the easy way to get "distiller's yeast" performance without
fooling about with yeast starters and the like.
There's an excellent book by James Hay called "Homespun Spirits", explaining
how to "spin" the alcohol out. Basically what is done is ...
Standard wash is prepared as for distillation - resulting in a liquor of 12-20% alcohol. This is then poured
into 1.25 - 2L PET soft-drink bottles. Fill the bottles with little headspace.
Shake the bottle to aerate them, so that plenty of air bubbles will form during
freezing. Freeze solid (may take several days). Cut V shaped slits into the side of the bottles.
so that the points face away from direction of rotation. Approx five rows of
four slits, each with sides & base approx 10mm long.
Make a couple of air holes above the solid surface. Modify a bottle cap
so that it fits into a power-drill - eg thread a long bolt through it, with
a couple of washers, and a nut to hold it all in place. Tighten the drill
chuck around the length of bolt that extends. With the bottle attached to
the drill, spin the bottle inside a tall container (rubbish bin).
Use a variable speed drill, so that you can control the speed.
The slits create an aerodynamic drag, forming a vacuum. This causes the
air bubbles to expand, breaking up the ice, and releasing the non-frozen alcohol.
Keep at full revs until no more alcohol is seen running down the side of the bucket, or ice
starts coming out the slits. Slow the speed down slowly.
Store the spirit for a couple of weeks to let it clear, then polish as for
distilled spirit.
However, Ups474 writes :
More correctly known as "fractional crystillization", the practice of
freezing an alcoholic mash then removing the (concentrated)alcohol
was used in ancient times before distilling was known. The problems
with trying to purify alcohol this way was that not only did the
ethanol come out of the block of ice, but so did all the nasty higher
and lower alcohols that cause painfull hangovers, and there is no way
to separate them from the ethanol by freezing.
The other reason you
may have already seen: if you have ever put a bottle of vodka in the
freezer (a traditional method of serving), you will notice that no
matter how long it is in there, it never does anything but turn
slightly thick, like syrup- no water freezes out once the alcohol
gets up to 40%. Unless you have easy access to a source of liquid
nitrogen, etc. so you can "super cool" the mash, then bring it's temp
back up in a controlled enviroment so as to melt off various alcohols
at various times, it's not worth bothering. Even then, it ain't
worth it-the energy needed to keep the mash that cold would exceed
the energy requirements needed to distill it.
Home winemakers use
this technique to make fortified wines without using distilled
spirits- Put the mash in a plastic jug, leaving enough room for
expansion, then put this in the freezer until it's a solid block of
ice, then invert the container over a collection jar and gather
everything that melts out until you have collected half of what is in
the jug. With a 10% mash, this will come out to about 17% ABV.
AppleJack
Here's what I've found on making Applejack. Its from a 1957 book
"Home-made wines" by Mary Aylett, you may find similar in other
wine-making books at your local library.
- Apple-jack.
Make a good strong cider, as described in the recipe given,
then put it in a sealed cask. Place the cask in some place
where it can freeze through very slowly. In Canada this is doen by
digging a large hole in the ground and covering the cask about three
inches deep. When the thaw comes, the cask is at once dug up, and
the contents will be found to have separated into apple water and to
pure spirit. The spirit will be in the centre of the cask. The ice
must be broken with great care, and the liquid spirit in the centre
carefully run off. This is the apple-jack. It should nver be drunk
undiluted.
- Cider.
Any apples will make good cider, but cider apples are best.
If sweet apples are used a mixture of bitter apples or crabs is
to be advised. Put the apples into a wooden tub, and smash them
roughly with a wooden balk until they are a crude pulp. Cover the
tub and leave for twenty four hours. Then put the pulp through a
press or crush it in a strong hessian bag. As the juice runs, put
it in a cask, and see that it is quite full. Keep the cask in a warm
place and allow it to ferment spontaneously which as a rule it does
very quickly. At a normal autumn temperature, the apples will ferment
within 48 hours. Top up the cask with apple juice until the first
violence of the ferment has subsided, then put the cask in a cool
place to finish. Do not bung down hard until all sounds of fermenta
tion has ceased. It will be ready to drink in three months, but will
be better if left for six. It will improve if well racked. For
sweet cider add sugar at the rate of a pound to a gallon, or more to
taste. (Through the book, Mary is a firm believer in natural fermentation.
I'd play on the side of caution, and direct the fermentation by
adding a suitable yeast.)
Air Stripping
Yet another technique is to strip the alcohol from the wash using air bubbles. A smaller
bucket of wash is put inside a larger bucket, with an airstone (like that used in a fish aquarium)
bubbling small bubbles up through the wash. The vapour will strip some alcohol out from the wash,
and it will condense on the walls of the outer container. I suspect that a fair amount gets lost too - as
some air will always be exiting the setup. Maybe you'd arrange it so that the air intake to the pump
was inside the buckets, so that you were just recycling the same air.

